Finalizing the results of a multi-year, multi-million dollar study into a single, cohesive document is a monumental task. The success of this final step hinges on a well-structured and comprehensive Clinical Trial Report Template. This document, formally known as a Clinical Study Report (CSR), is not merely a summary of findings; it is the definitive, integrated record of a clinical trial’s conduct and results. It serves as the primary basis for regulatory agencies like the Food and Drug Administration (FDA) and the European Medicines Agency (EMA) to evaluate a new drug or medical device’s safety and efficacy. Without a standardized framework, vital information could be misplaced, misinterpreted, or omitted, jeopardizing the entire submission process.
The complexity of a CSR cannot be overstated. It must meticulously detail every aspect of the trial, from the initial scientific rationale and study objectives to the statistical analysis methods and the final interpretation of the data. This report weaves together the study protocol, patient data, safety assessments, and efficacy outcomes into a single, coherent narrative. For regulatory reviewers, who must scrutinize vast amounts of information to make critical public health decisions, a predictable and logical structure is essential. It allows them to navigate the report efficiently, locate specific data points, and verify the integrity of the trial’s execution and conclusions.
This is precisely why a robust template is not just a convenience but a necessity in clinical research. It provides the essential scaffolding for medical writers, statisticians, and clinicians to build a compliant and high-quality report. By following a globally accepted standard, research organizations can ensure consistency across their development programs, streamline the writing and review process, and significantly improve their chances of a successful regulatory submission. This article will delve into the critical components of a clinical trial report, the international guidelines that govern its structure, and the best practices for utilizing a template to produce a clear, complete, and compelling CSR.
Understanding the Clinical Study Report (CSR)
A Clinical Study Report (CSR) is the cornerstone of any regulatory submission for a new therapeutic product. It is an extensive, stand-alone document that presents a complete and unabridged account of a single clinical trial. Unlike a journal publication, which is a condensed summary for a broad scientific audience, the CSR is created for a highly specialized audience: regulatory reviewers. Its purpose is to provide them with all the necessary information to independently re-evaluate the study’s findings and confirm the sponsor’s conclusions regarding the investigational product’s safety and effectiveness.
The scope of a CSR is all-encompassing. It integrates clinical and statistical descriptions, presentations, and analyses into a single report. It includes detailed information on the study protocol, patient demographics, how the treatment was administered, the methods used for data collection, and the complete statistical analysis plan. Furthermore, it must present a transparent account of all patients, including those who withdrew from the study, and provide a thorough evaluation of all adverse events. The CSR is supported by a massive volume of appendices that can include the full study protocol, sample case report forms (CRFs), investigator information, and extensive patient data listings.
Ultimately, the CSR serves as a critical piece of evidence in the benefit-risk assessment of a new drug or device. It must be written with absolute clarity, precision, and objectivity. Every claim made in the report must be traceable to the source data contained within its tables, listings, and figures. A well-written CSR tells the complete story of the trial, including its successes, challenges, and limitations, thereby enabling regulators to make a fully informed decision about its approval.
The Foundation: ICH E3 Guideline for Structuring a CSR
The global standard for the structure and content of a CSR is laid out in the International Council for Harmonisation (ICH) E3 guideline. The ICH brings together regulatory authorities and the pharmaceutical industry to discuss scientific and technical aspects of drug registration. Its guidelines are designed to ensure that safe, effective, and high-quality medicines are developed and registered in the most efficient and ethical manner. The ICH E3 guideline is the universally accepted framework that dictates how a CSR should be organized, ensuring consistency and completeness for regulatory submissions across Europe, Japan, the United States, and other regions.
The ICH E3 guideline provides a detailed “table of contents” for the CSR, breaking it down into 16 distinct sections. This standardized format is crucial because it creates a predictable structure that reviewers can easily navigate. They know exactly where to find information on the study’s design, patient disposition, efficacy results, or safety data, regardless of which company sponsored the trial. Adherence to this guideline is not optional for major regulatory submissions; it is a fundamental requirement.
Using the ICH E3 structure as the basis for a Clinical Trial Report Template ensures that all necessary components are included from the outset. This minimizes the risk of receiving a “Refuse to File” letter from a regulatory agency due to an incomplete or poorly organized report, which can cause significant delays and financial losses. The guideline provides the blueprint for a report that is not only compliant but also logical, transparent, and review-friendly.
Key Components of a Clinical Trial Report Template
A robust clinical trial report template is built directly upon the framework provided by the ICH E3 guideline. Each section has a specific purpose and requires detailed, accurate information.
Title Page
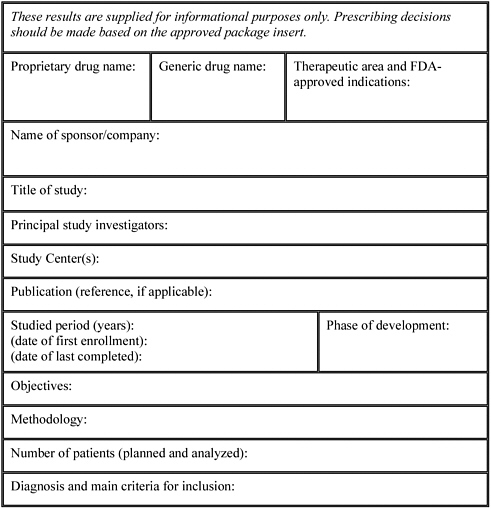
The very first page must clearly identify the study. It includes the study title, the name of the investigational product, the indication studied, the name of the sponsor, the protocol identification number, the development phase (e.g., Phase I, II, III), the study start and end dates, and the date of the report.
Synopsis
The synopsis is a brief (typically 1-3 pages) but comprehensive summary of the study. It must stand on its own and provide a concise overview of the trial’s design, objectives, patient population, methodology, key results, and conclusions. Reviewers often read the synopsis first to get a high-level understanding of the study before diving into the main body of the report. It needs to be written with extreme clarity and accuracy.
Table of Contents
A detailed Table of Contents is required to help reviewers navigate the extensive document. It should list all sections, subsections, tables, figures, and appendices with their corresponding page numbers.
Introduction
This section sets the stage for the study. It should briefly describe the investigational drug, the medical condition it is intended to treat (the indication), and the scientific rationale for conducting the trial. It summarizes the relevant preclinical and clinical data that supported the decision to proceed with this particular study.
Study Objectives
Here, the primary and secondary objectives of the trial must be clearly and explicitly stated. The primary objective is the main question the study was designed to answer and is directly tied to the primary efficacy endpoint. Secondary objectives relate to other effects of the treatment that the study was designed to evaluate.
Investigational Plan
This is one of the most substantial sections of the report. It details the “how” of the study and is often broken down into several sub-sections:
* Overall Study Design and Plan: A description of the trial type (e.g., randomized, double-blind, placebo-controlled, parallel-group) and a schematic diagram illustrating the design, procedures, and stages.
* Study Population: A detailed description of the inclusion and exclusion criteria used to select patients for the trial.
* Treatments: Precise information on the treatments administered, including the investigational product and any comparators (placebo or active control). It specifies the dose, route of administration, frequency, and duration of treatment for each group.
* Efficacy and Safety Assessments: This lists all the efficacy and safety variables that were measured, the methods used to measure them, and the schedule of assessments (i.e., when they were performed).
* Statistical Methods: This sub-section describes the statistical plan for analyzing the data, including the sample size calculation, definitions of analysis populations (e.g., Intent-to-Treat), statistical tests used for primary and secondary endpoints, and procedures for handling missing data.
Study Patients
This section provides a complete accounting of the patients in the trial. It includes a section on patient disposition, which tracks the flow of patients from screening through to study completion, detailing how many were randomized, received treatment, completed the study, and discontinued (along with the reasons why). It also presents detailed demographic and baseline characteristics of the study population.
Efficacy Evaluation
Here, the results related to the study’s objectives are presented. This section includes detailed analyses of the primary and secondary efficacy endpoints. The data is typically presented through a combination of text, tables, and figures, with a clear focus on demonstrating whether the study met its pre-specified objectives.
Safety Evaluation
A thorough assessment of safety is a critical component of every CSR. This section details the extent of patient exposure to the drug, followed by a summary of all adverse events (AEs), serious adverse events (SAEs), and deaths. It also presents analyses of clinical laboratory data, vital signs, and other safety measurements.
Discussion and Overall Conclusions
In the discussion, the efficacy and safety results are summarized and interpreted in the context of the study’s objectives. It should provide a critical assessment of the study’s limitations and the implications of the findings. The conclusion presents a clear benefit-risk assessment of the investigational product based on the trial data.
Appendices
The appendices contain a vast amount of supplementary information and source documentation. Common appendices include the study protocol, sample case report forms, investigator-related information, statistical analysis plan, patient data listings, and regulatory documentation like Institutional Review Board (IRB) approvals.
Why Using a Standardized Template is Crucial
Adopting a standardized template based on ICH E3 is a strategic imperative for any organization involved in clinical research. The benefits extend far beyond simply meeting a regulatory requirement.
First and foremost is regulatory compliance. A well-constructed template acts as a checklist, ensuring that all 16 required sections and their sub-components are addressed. This drastically reduces the risk of an incomplete submission, which can lead to costly delays in the drug approval timeline.
Second, it promotes consistency and quality. When all teams within an organization use the same template, it ensures a uniform structure and level of detail across different studies. This creates a portfolio of high-quality, professional documents that are easier to manage, review internally, and compile for larger submissions like a New Drug Application (NDA).
Third, a template drives efficiency. It provides a clear roadmap for the medical writing team, eliminating the need to design the report’s structure from scratch for every new study. This allows writers to focus their efforts on accurately and clearly presenting the scientific data and narrative, rather than on formatting and organization.
Finally, a standardized structure enhances clarity and reviewability. For both internal reviewers and external regulatory authorities, a predictable format makes it significantly easier to navigate the document and find specific information. This facilitates a more efficient and thorough review process, which is beneficial for all stakeholders.
Common Pitfalls to Avoid
While a template is an invaluable tool, it can be misused. Simply “filling in the blanks” without careful thought can lead to a low-quality report. One common pitfall is the overuse of “boilerplate” language. The report must be tailored to the specific study; a generic description of a statistical method or study design that doesn’t reflect the actual protocol can create confusion and undermine credibility.
Another major issue is failing to address discrepancies. No clinical trial is perfect. There will always be deviations from the protocol. The CSR must transparently describe these deviations and analyze their potential impact on the study’s results. A template should not be used to gloss over these important details.
Incomplete or poorly written safety narratives are another red flag for reviewers. For every death or serious adverse event, a detailed narrative is required. This narrative should provide a clear, concise medical story of the event, and a template should only serve as a guide for the required content, not as a substitute for careful clinical interpretation.
Finally, the report must tell a cohesive story. The data presented in the tables and figures must align with the interpretations in the text. The discussion and conclusion must logically follow from the results presented. A template provides the structure, but the medical writer must provide the coherent narrative that ties it all together.
Conclusion
A Clinical Trial Report Template, built on the robust foundation of the ICH E3 guideline, is an indispensable asset in the field of clinical development. It is far more than a simple document format; it is a strategic tool that ensures regulatory compliance, enhances quality, and improves efficiency. By providing a standardized and logical structure, the template allows the entire clinical team to collaborate effectively in producing a comprehensive and clear account of a trial’s conduct and findings. This detailed report is the ultimate deliverable of a clinical study, forming the basis for critical decisions that impact patient health worldwide. While the template provides the essential framework, it is the meticulous attention to detail, scientific integrity, and clear narrative writing that transforms it into a successful Clinical Study Report, capable of withstanding the intense scrutiny of regulatory review and paving the way for new medical innovations.
]]>